Introduction:
Peripheral T cell lymphomas (PTCLs) are a group of rare non-Hodgkin's lymphomas. The first-line standard therapy is CHOP or CHOP-like regimens, and ~50-65% patients could achieve tumor response. However, approximately 40% of patients with complete response and 80% of patients with partial response had disease relapse within 2 years after initial tumor response, and the prognosis of relapsed patients was very poor. There is a lack of standard maintenance therapy for this group of patients. Golidocitinib is a JAK1 selective inhibitor which showed promising antitumor efficacy in patients with relapsed or refractory PTCLs. Here we report the antitumor efficacy and safety of golidocitinib as maintenance treatment after first-line therapy in a phase 2 clinical trial (JACKPOT26, CTR20213318).
Methods:
PTCL patients who achieved tumor response post first-line therapy were enrolled and received golidocitinib treatment at 150 mg once daily (QD). This study included two cohorts: cohort 1 (complete response post first-line therapy) and cohort 2 (partial response post first-line therapy). The objectives were to assess safety and antitumor efficacy of golidocitinib as maintenance therapy. The efficacy endpoints included the rate of patients who were 1-year and 2-year disease free survival (DFS) for cohort 1, and progression free survival (PFS), objective response rate (ORR), duration of response (DoR) for cohort 2, respectively. Tumor response was assessed by investigators based on CT images per Lugano 2014 criteria. Endpoints for safety assessments are treatment emergent adverse events (TEAEs) and serious adverse events (SAEs), etc.
Results:
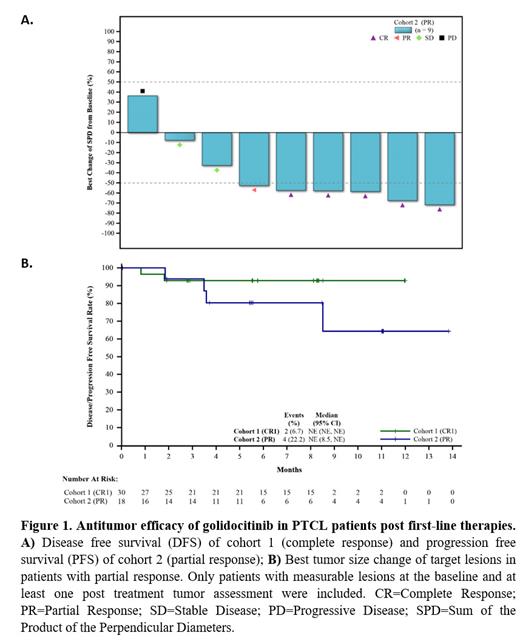
A total of 48 patients with PTCL who had achieved tumor response post first-line treatment were enrolled, including 30 patients with complete response (cohort 1) and 18 patients with partial response (cohort 2). The median age was 58.5 years (range: 25 to 74), and 52.1% were male. Major pathological subtypes included: AITL (31.3%), NOS (27.1%) and NK/TCL (27.1%). All dosed patients were included in the efficacy and safety analysis.
As of June 27, 2023, the median duration on treatment for all patients was 7.1 months. In cohort 1, with median follow-up time of 8.1 months, median DFS has not been reached, with 86.7% patients still responding. In nodal subtypes(AITL, NOS, ALK-ALCL), with median follow-up time of 8.3 months, median DFS has not been reached, with 82.4% patients still responding. In cohort 2, with median follow-up time of 5.6 months, median PFS has not been reached, with 66.7% patients still responding. The ORR of cohort 2 was 38.9%, and complete response rate (CRR) was 33.3%. In 10 patients with measurable disease at the baseline, six patients (60%) achieved tumor response, with 5 patients achieved complete response. With medina follow-up time of 3.7 months, the median DoR has not been reached, with 85.7% of patients still responding. The estimated 6-month and 9-month DoR rates were 100% and 100%, respectively.
The safety profile of golidocitinib was similar as what has been reported previously. A total of 32 patients (66.7%) reported ≥ grade 3 drug-related TEAEs. The most common ≥ grade 3 drug-related TEAEs were hematological adverse events in nature, including neutropenia (37.5%), leukopenia (31.3%), lymphopenia (10.4%) and thrombocytopenia (6.3%). The majority of these TEAEs were reversible and clinically manageable. Only one patient discontinued golidocitinib treatment due to drug-related TEAEs. No drug-related TEAEs leading to fatal outcome was reported.
Conclusions:
In conclusion, golodocitinib showed manageable safety profile and promising effect in maintaining and enhancing tumor response in patients with PTCL post first-line therapies. This result supports further clinical development of golidocitnib in the first-line setting. The updated data will be presented in the conference.
Disclosures
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal